Back Topic |
Go to Index Class-XI |
Next Topic |
Micro-Bio-Molecules:
1. Sugars
INDEX
- Sugars: An Introduction
- Monosaccharides: Chemical Properties
- Chemical Composition
- Functional Group
- Aldoses Sugars
- Ketoses Sugars
- Functional Group
- Reduction Reaction
- Non Reducing Sugars
- Reducing Sugars
- Chemical Composition
- Nomenclature & Structural Configurations
- D/L Form & d/l Form of Sugars
- Chain & Ring Forms of Sugars
- Acyclic (Fischer's Projection)
- Cyclic (Howarth Projection)
- Hemiacetal & Hemiketal
- Furanose & Pyranose
- Alpha & Bita Positions
- Anomeric Sugars
- Alpha & Bita Positions
- Epimeric Sugars
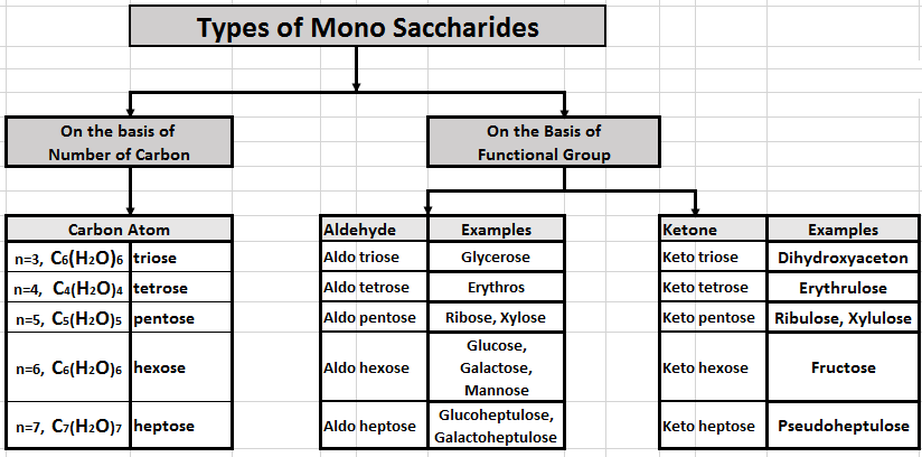
- Types of Sugars (MonoSaccharides)
- On the basis of Functional Group
- On the Basis of Number of Carbons
- On the Basis of Aromaticity (Cyclic & Acyclic Form)
- On the Basis of D/L & d/l Form of Sugars
- On the basis of Reduction Reaction
- Detection Techniques of Sugars (MonoSaccharides)
- For Non Reducing & Reducing Sugars
- For Anomeric Sugars (Fructose & Glucose)
Sugars: An Introduction
- "Sugar" term is derived from Sanskrit शर्करा śarkarā,
- Also known as "saccharides"
- Is organic compound, Generalized name for sweet, short-chain, soluble carbohydrates.
- Composed of carbon, hydrogen, and oxygen.
- Simple sugars are called monosaccharides.
- The formula "Cn(H2O)n" (n is between 3 and 7).
- The names of typical sugars end with -ose, such as "glucose", "dextrose",
- Is Polyhydroxyaldehyde or Polyhydroxyketone Carbon Chain / Cycle, Contain either
- aldehyde (-CHO) groups is called as aldoses
- ketone (-C=O) groups. is called as ketoses
- Monosaccharaides are the most basic units of Sugars.They are the simplest form of sugar and are usually colorless, water-soluble, crystalline solids.
MonoSaccharides: Chemical Properties
- Monosaccharaides are the most basic units of Sugars.
- They are the simplest form of sugar and are usually colorless, water-soluble, crystalline solids.
- They cannot be further hydrolised to simpler compounds.
- The general formula is {(Cn H2n On)} where n is 1, 2, 3 .... .
A. Chemical Composition
- With few exceptions (e.g., deoxyribose), monosaccharides have this chemical formula: Cx(H2O)y, where conventionally x ≥ 3.
- Monosaccharides can be classified by the number x of carbon atoms they contain:
- diose (2 carbons)
- triose (3 carbons)
- tetrose (4 carbons),
- pentose (5 carbons),
- hexose (6 carbons),
- heptose (7 carbons) - mannoheptulose and sedoheptulose, and so on.
- The most important monosaccharide, glucose, is a hexose.
- Monosaccharides with eight or more carbons are rarely observed as they are quite unstable.
- aldehyde (-CHO) groups is called as aldoses
- ketone (-C=O) groups. is called as ketoses
B. Reduction Reaction
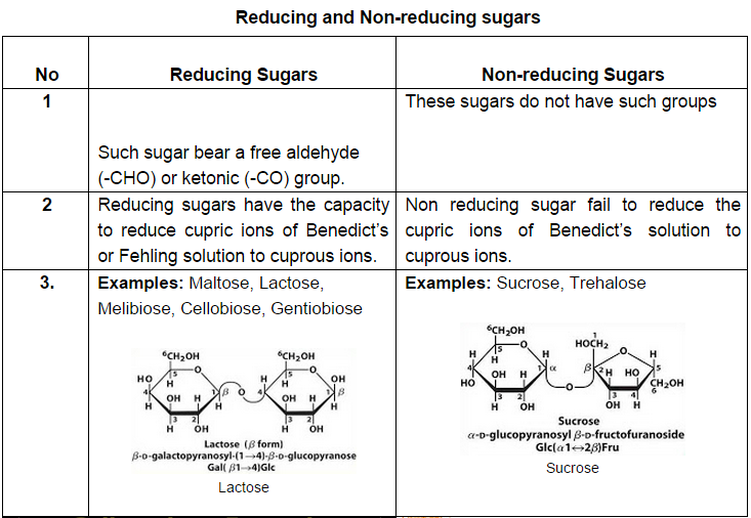
1. Reducing Sugars-
- any sugar that is capable of acting as a reducing agent
- because it has a free aldehyde group or a free ketone group.
- Allmonosaccharides are reducing sugars, along with some disaccharides,oligosaccharides, and polysaccharides.
- The monosaccharides can be divided into two groups:
- the aldoses, which have an aldehyde group, and
- the ketoses, which have a ketone group. Ketoses must first tautomerizeto aldoses before they can act as reducing sugars.
- The common dietary monosaccharides galactose, glucose and fructose are all reducing sugars.
- for example in the Tollens' test orBenedict's test.
2. Non-Reducing Sugars-
- A nonreducing sugar is not oxidized by a weak oxidizing agent (an oxidizing agent that oxidizes aldehydes but notalcohols, such as the Tollen’s reagent) in basic aqueous solution.
- The characteristic property of nonreducing sugars is that, in basic aqueous medium, they do not generate any compounds containing an aldehyde group.
- eg: sucrose, which contains neither a hemiacetal group nor a hemiketal group and, therefore, is stable in water.
Differences Between Reducing & Non-Reducing Sugars
Characterization of Reducing & Non-Reducing Sugars
- A qualitative tests are used to detect the presence of reducing sugars.
- Two of them use solutions of copper(II) ions:
- Benedict's reagent (Cu2+ in aqueous sodium citrate) and
- Fehling's solution (Cu2+ in aqueous sodium tartrate).
- Tollen's reagent (Ag+) in aqueous ammonia.
- The reducing sugar reduces the copper(II) ions in these test solutions to copper(I), which then forms a brick red copper(I) oxide precipitate.
- Reducing sugars can also be detected with the addition of Tollen's reagent, which consist of silver ions (Ag+) in aqueous ammonia. When Tollen's reagent is added to an aldehyde, it precipitates silver metal, often forming a silver mirror on clean glassware.
Nomenclature & Structural Configurations
A. D/L Form & d/l Form of Sugars
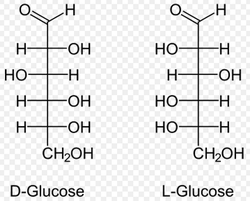
- are identified with the prefixes D- and L-, according to the sense of rotation:
- D- is dextrorotatory (rotates the polarization axis clockwise), while
- L- is levorotatory (rotates it counterclockwise).
- Note that the D- and L- prefixes do not indicate the direction of rotation of polarized light, which is a combined effect of the arrangement at all chiral centers. However, the two enantiomers will always rotate the light in opposite directions, by the same amount.
B. Chain & Ring Forms of Sugars
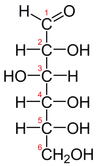
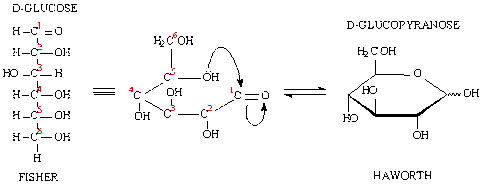
1. Acyclic (Fischer's Projection)
- The Fischer projection is a systematic way of drawing the skeletal formula of an acyclic monosaccharide
- In the Fischer projection, two mirror-image isomers differ by having the positions of all chiral hydroxyls reversed right-to-left.
- Mirror-image isomers are chemically identical in non-chiral environments, but usually have very different biochemical properties and occurrences in nature.
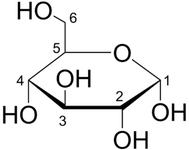
2. Cyclic (Howarth Projection)
- form through a nucleophilic addition reaction between
- the carbonyl group (-C=O) and
- one of the hydroxyls (-OH) of the same molecule.
- creates a ring of carbon atoms closed by one bridging oxygen atom.
- The resulting molecule has
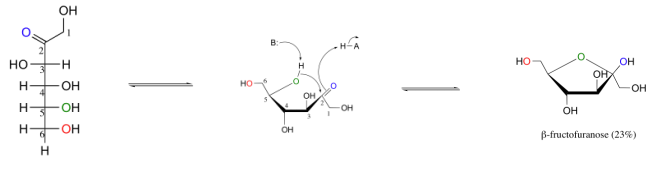
a. Hemiacetal & Hemiketal
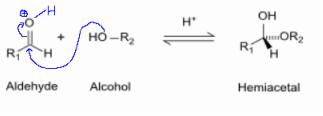
1. Hemiacetal (Product of "-CHO" + "-OH")
- an hemiacetal or
- Reaction between Aldehyde (-CHO) and Hydroxyl group (-OH)
- Found in Ketoses (mostly in KetoPentoses (5C) & KetoHexose (6C).
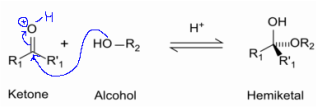
2. Hemiketal (Product of "-C=O" + "-OH")
- hemiketal group,
- Reaction between Ketone (-C=O) and Hydroxyl group (-OH)
- Found in Ketoses (mostly in KetoPentoses (5C) & KetoHexose (6C).
b.Furanose & Pyranose
In these cyclic forms, the ring usually has
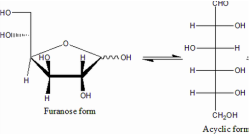
1. Furanose (5C membered Hemiacetal Structure)
- with a 5-Carbon membered ring structure, called glucofuranose.
- form hemiketal linkage reaction take place between
- 1st carbons (-C=O) ketone group and
- 4th Carbon (-OH) Hydroxyl Group
- form hemiketal linkage reaction take place between
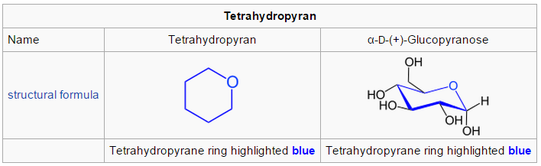
2. Pyranose (6C membered Hemiketal Structure)
with 6-carbons membered molecule is the aldohexose glucose called glucopyranose.
with 6-carbons membered molecule is the aldohexose glucose called glucopyranose.
- may form a hemiacetal linkage between
- the hydroxyl on carbon 1 and
- the oxygen on carbon 4,
C. Anomeric Sugars
Alpha & Bita Form
- they are a class of stereoisomer called an anomer.
- Anomers are capable of interconverting in solution. All cyclic structures of monosaccharides exhibit anomeric α (down) andβ (up) versions.
- These differences occur at the anomeric acetal carbon (the only carbon with two C−O bonds.
α- form
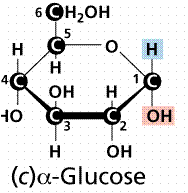
- In the α- form, the exocyclic O group at the anomeric center is on the opposite face to the -CH2OH group.
- The Alpha position is defined as the -OH being on the opposite side of the ring as the C # 6. In the Haworth structure this also results in a downward projection.
- The Alpha position is defined as the -OH being on the opposite side of the ring as the C # 6. In the chair structure this results in a downward projection.
β- form
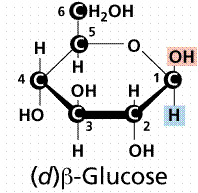
- In the β- form, the exocyclic O group at the anomeric center is on the same face as the -CH2OH group.
- The Beta position is defined as the -OH being on the same side of the ring as the C # 6. In the Haworth structure this results in an upward projection.
- The Beta position is defined as the -OH being on the same side of the ring as the C # 6. In the chair structure this results in a horizontal projection.
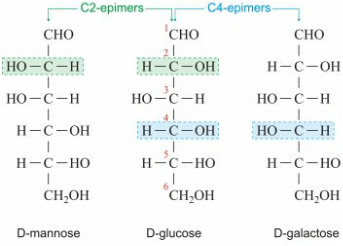
D. Epimeric Sugars
- epimer refers to one of a pair of stereoisomers
- The two isomers differ in configuration at only one stereogenic center.
- In glucose, the -OH group on the first carbon is in the axial position, the direction opposite the -OH group on carbon C-4.In galactose, the -OH group is oriented in the same direction, the equatorial position.
Types of MonoSaccharides Sugars
Characterization Techniques of Sugars (MonoSaccharides)
1. For Non Reducing & Reducing Sugars
- A qualitative tests are used to detect the presence of reducing sugars.
- Two of them use solutions of copper(II) ions:
- Benedict's reagent (Cu2+ in aqueous sodium citrate) and
- Fehling's solution (Cu2+ in aqueous sodium tartrate).
- Tollen's reagent (Ag+) in aqueous ammonia.
- The reducing sugar reduces the copper(II) ions in these test solutions to copper(I), which then forms a brick red copper(I) oxide precipitate.
- Reducing sugars can also be detected with the addition of Tollen's reagent, which consist of silver ions (Ag+) in aqueous ammonia. When Tollen's reagent is added to an aldehyde, it precipitates silver metal, often forming a silver mirror on clean glassware.
2. For Anomeric Sugars (Fructose & Glucose)
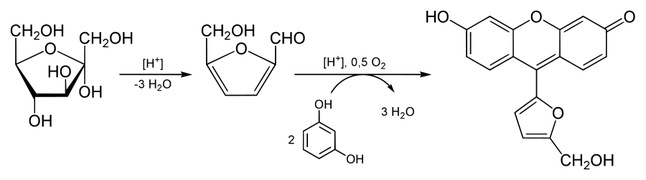
Seliwanoff’s test
- Seliwanoff’s test is a chemical test which distinguishes between aldose and ketose sugars.
- This test is based on the fact that, when heated, ketoses are more rapidly dehydrated than aldoses. It is named after Theodor Seliwanoff, the chemist that first devised the test.
- The reagents consist of resorcinol and concentrated hydrochloric acid:
- The acid hydrolysis of polysaccharides and oligosaccharides yields simpler sugars followed by furfural.
- The dehydrated ketose then reacts with the resorcinol to produce a deep cherry red color. Aldoses may react slightly to produce a faint pink color.
- Fructose and sucrose are two common sugars which give a positive test. Sucrose gives a positive test as it is a disaccharide consisting of fructose and glucose.